INTRODUCTION
Treatment of immune thrombotic thrombocytopenic purpura (iTTP) includes plasma exchanges (PEX) and immunosuppressive treatment with steroids with or without rituximab (RTX). Actually, caplacizumab (CPZ) has been included in this scheme of treatment, because of its effect in decreasing clinical response time, exacerbations and refractoriness. A longer time to recovery of ADAMTS13 levels has recently been described in a group of patients from United Kingdom in subjects treated with caplacizumab. We analysed the behaviour of ADAMTS13 recovery in patients from the Spanish TTP registry (REPTT).
METHODS
Retrospective, multicentre, non-interventional study. Data collected by the Spanish PTT Registry (REPTT) from September-2018 to February-23. The objective is to describe the efficacy of standard therapy with and without CPZ with regard to recovery of ADAMTS13 levels higher than 20%. Episodes evaluable for ADAMTS13 >20% recovery were those with data available 48 hours or more after last PEX. Data about clinical response, clinical remission, exacerbations and relapses will be described. Qualitative variables described in percentages, quantitative in median and interquartile range (IQR).
RESULTS
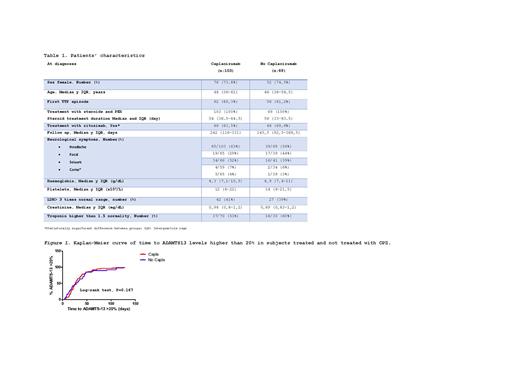
We included 178 patients from REPTT, but only 164 were analysed. Reasons for exclusion were 5 were congenital TTP, 3 patients were younger than 16 years-old, and 6 patients because of lack of data. The 164 patients presented 172 episodes whose characteristics are described in Table 1. Clinical remission rate did not differ between groups, 95% CPZ vs 97% no CPZ, with a median time of 41 days (36-50) vs 43 days (38-50), respectively. A median of 11 PEX (IQR:6-20) were required to get clinical remission, lower in CPZ group (CPZ 10 PEX, IQR:5-18 vs no CPZ 12 PEX, IQR: 7-23, p=0.044). In 49 of the 103 CPZ episodes, CPZ was introduce in the 3 first days from diagnosis (CPZ3). In this group, number of PEX was critically reduced (CPZ 6 PEX, IQR: 4-9 vs no CPZ 12 PEX, IQR: 7-23; p<0.001). Exacerbation rate was lower CPZ group (12.5% vs 23.2%, p. 0.033), as refractoriness rate(4.1% vs 8.7%, p = 0.038).
With a median follow-up of 188.5 days (110.5-354.5), relapse rate was 10.5%, lower in subjects treated with CPZ (4% vs 25.5%, p < 0.001). Mortality before reaching clinical remission was 1.9% in the CPZ group and 4.3% in the non-CPZ group (p. 0.177). Adverse events (AEs) rate was 10.5% in the non-CPZ group, most related to plasma infusion, all mild. Incidence of AEs related to CPZ was 24.5%, with 12 bleeding events: 11 WHO grade 1 (3 metrorrhagia, 2 pericatheter bleeds, 6 mucosal bleeds) and one grade 2 (1 gastrointestinal in a patient with intestinal polyposis).
Regarding patients reaching ADAMTS13 >20%, we found no differences with regard to CPZ use (Fig 1). There were 114 evaluable episodes (66% of serie): 39 No-CPZ (56.5% of No CPZ group) and 75 CPZ group (72.8% CPZ group). The 93% achieved ADAMTS13 levels >20%, with no difference between groups(CPZ 93.3% vs No CPZ 94.9%, p. 0.082). In the 107 episodes who reached ADAMTS13 >20%, time from start of PEX to ADAMTS13 > 20% was 27 days (IQR:17-51),(CPZ 26.5 days, IQR:17.75-39, vs no CPZ 22 days, IQR:14.75-33.5, p.0.077). ADAMTS13 levels at that time were 51% (IQR: 36-80%),(CPZ 50%, IQR: 33-71% vs No CPZ 59%, IQR: 37-89%, p. 0.437). Time from last PEX to ADAMTS13 levels >20% was similar in both groups (CPZ 15.5 days, IQR: 7.75-29.25 vs No CPZ 13 days, IQR: 8-22 days, p. 0.063). In patients treated with CPZ in the 3 first days from diagnosis, there is no difference in time from start PEX and ADMATS13 >20%, but we found longer time from last PEX to ADAMTS13 >20% (CPZ3 18 days, IQR: 12-43 days vs No CPZ 13 days, IQR: 8-22 days, p. 0.026). The 81.3% of the episodes who reached ADAMTS13 levels >20% received RTX and 71.4% (2/7) of the non-responders (p. 0.385). We found no differences in time to ADAMTS13 > 20% in none of both groups with regard of RTX used.
CONCLUSIONS
CPZ is safe and effective in the treatment of iTTP. Its use in the first 3 days of diagnosis shortens clinical response times. Exacerbation and refractoriness are lower in patients treated with CPZ.
In our series, the use of CPZ did not extend time to ADAMTS13 levels greater than 20%, neither from the start of PEX nor from PEX stop evaluation. Only in the group of patients treated with CPZ in the first 3 days of diagnosis, time from last PEX to ADAMTS13 >20% is longer, probably because they need less PEX, but time from PEX start to ADAMTS13 >20% is the same than in the rest of patients.
Disclosures
De La Rubia:Menarini: Honoraria; Takeda: Research Funding; Sanofi: Speakers Bureau; Pfizer: Speakers Bureau; Janssen: Honoraria, Speakers Bureau; GSK: Honoraria, Research Funding, Speakers Bureau; BMS: Honoraria; Oncopharm: Honoraria. Valcárcel:BMS/Celgene: Consultancy, Other: Travel expense reimbursement, Speakers Bureau; Amgen: Consultancy, Other: travel expense reimbursement, Speakers Bureau; GSK: Consultancy, Other: Travel expense reimbursement; Novartis: Consultancy, Other: Travel expense reimbursement, Speakers Bureau; Takeda: Consultancy; Pfizer: Consultancy, Other: Travel expense reimbursement, Speakers Bureau; Sanofi: Consultancy, Speakers Bureau; Jazz Pharmaceuticals: Consultancy, Other: Travel expense reimbursement, Speakers Bureau; SOBI: Consultancy; Agios: Speakers Bureau; Astellas Pharma: Speakers Bureau; Kyte: Speakers Bureau; Gebro Pharma: Speakers Bureau.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal